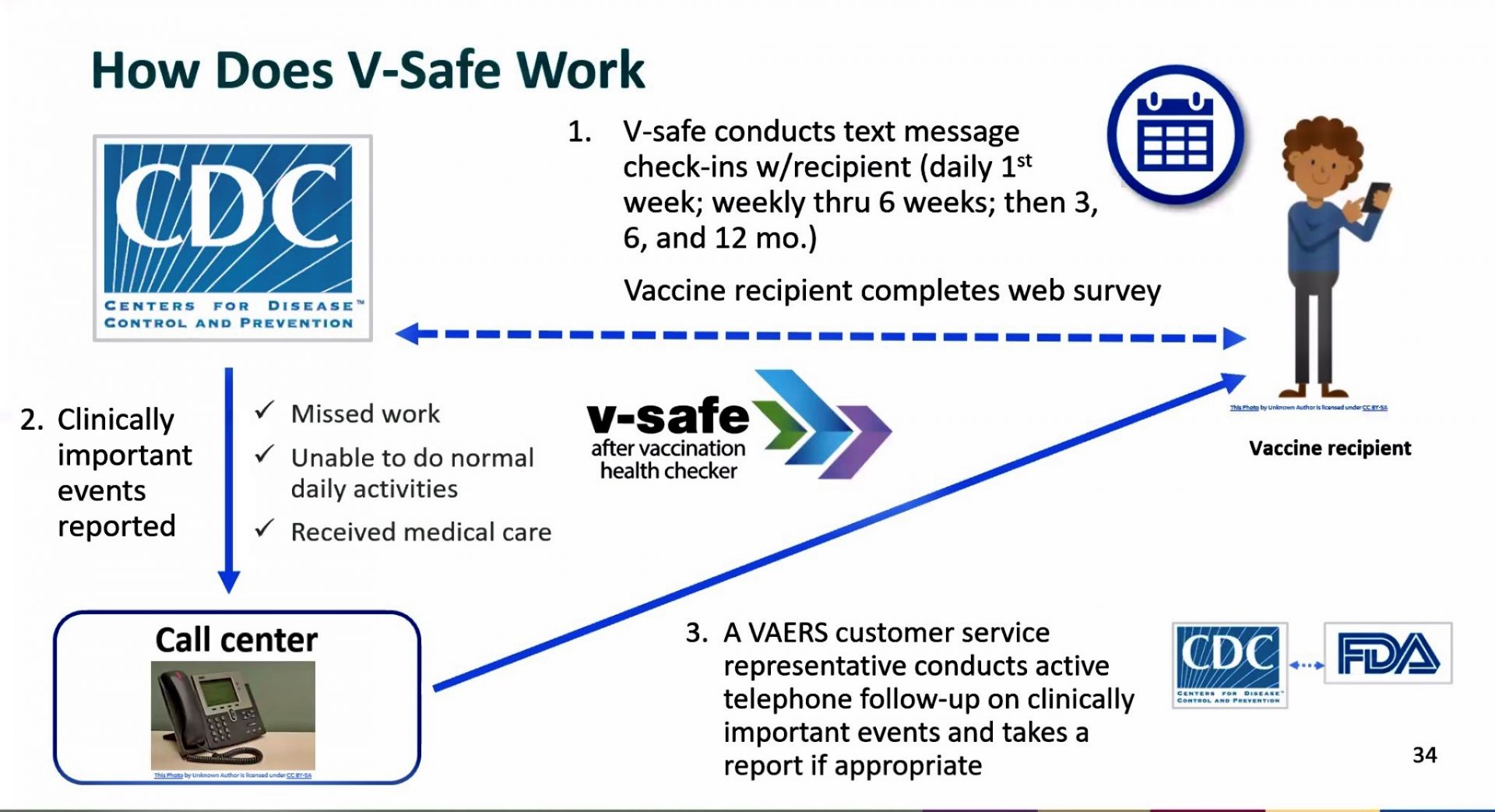
Learn more: How VAERS helps to monitor vaccine safetyĬDC and FDA currently have four existing safety monitoring systems in place to capture information about vaccination during pregnancy and closely monitors that information reported to each system.CDC also encourages reporting of any medically important adverse event even if it isn’t clear that the vaccine caused the health problem.  
For healthcare providers: Under Emergency Use Authorization, FDA requires healthcare professionals to report to VAERS certain adverse events that occur after COVID-19 vaccination.If you need further assistance reporting to VAERS, please email call 1-80. There are two ways to report an adverse event to VAERS: report online or report using a writable pdf form.The majority of reports to VAERS after COVID-19 vaccination have been non-serious adverse events.Reports of adverse events that are unexpected, appear to happen more often than expected, or have unusual patterns are further assessed. CDC and FDA’s Vaccine Adverse Event Reporting System (VAERS) - A national system that accepts reports from healthcare professionals, vaccine manufacturers, and the public of adverse events that happen after vaccination.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
